Effects of Acid Rain, Its Most Important Effects on Human
Effects of Acid Rain on Humans
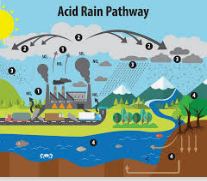
What is Acid Rain?
Air pollution such as sulfur dioxide and nitrogen oxides can cause or exacerbate respiratory diseases. Respiratory diseases made so much difficult to breath
Formation of Acid Rain
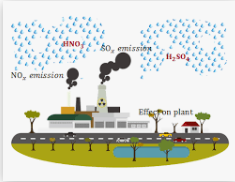
Sulfur dioxide and nitrogen dioxide and then water produce acids and nitric acid. The following answers will explain the acid reaction:
2SO2 (g) + O2 (g) + 2H2O (l) → 2H2SO4 (aq)
4NO2 (g) + O2 (g) + 2H2O (l) → 4HNO3 (aq)
Also Read:
- Eco-Friendly Energy
- Habitat, Component and Types of Habitat
- Disaster Management, Prevention and Mitigation
Effects of Acid Rain on Ecosystems
An environment is a collection of plants, animals, and other animals and their environment, including air, water, and soil. In nature everything is connected. If one part of the environment – plants or animals, soil or water – is contaminated, it can affect everything else.

Effects of Acid Rain on Fish and Wildlife
The environmental impact of acid rain is evident in aquatic environments such as rivers, lakes and wetlands where it can harm fish and other animals. Acid rain can pass through the ground and cause aluminum to leak from clay and then into rivers and lakes. The more acid that enters the ecosystem, the more aluminum is released.
Some plants and animals tolerate high levels of acid and aluminum. But some react to the acid and lose it when the pH drops. In general, juveniles of most species are more sensitive to environmental conditions than adults.
At pH 5, most fish do not lay eggs. When the pH is low, some large fish die. Some acid lakes do not have fish. While some fish or animals may tolerate slight acidity, this may not be the case for the animals or plants that eat them. Frogs, for example, have a pH of approx. 4, but fleas that bite them are more sensitive and may not stay below pH 5.5.
Effects of Acid Rain on Plants and Trees
Dead or dying plants are often found in acid-contaminated areas. Acid rain-dead or dying plants are often found in acid-contaminated areas. Acid rain causes aluminum to come out of the soil. This aluminum can harm plants and animals. Acid rain removes minerals and nutrients from the soil where plants need to grow.
At high levels, acid mist and clouds can remove nutrients from the leaves and leave brown leaves or needles. Trees then lose their ability to absorb sunlight, making them weaker and less sensitive to frost. This aluminum can harm plants and animals. Acid rain removes minerals and nutrients from the soil where plants need to grow.

Buffering Capacity
Many forests, rivers and lakes affected by acid rain remain unaffected because the soil in these areas can be neutralized by acidification of the waste and acidification can be prevented. This capacity depends on the weight, the composition and the soil type. In places like the mountains of the northeastern United States, the soil is soft and the water is acidic, so neutralization is not enough. Because these areas are particularly vulnerable, acids and aluminum accumulate in soils, rivers and lakes.
Episodic Acidification
Melting snow and heavy rainfall can cause episodic acidification. The lakes, which are generally free of high acidity, can temporarily experience the effects of rain as the snow melts or rains as the acid deposits rise and the soil becomes unsafe. This short-term growth in acidity (i.e. low pH) can cause short-term stress in an environment where organisms or species can be destroyed or killed.
Effects of Acid Rain on Materials
Most acidic substances are not impure. Sometimes soil particles can also become acidic and are called dry deposits. When acid rain and dry acidic particles fall to the ground, the nitric and sulfuric acids that acidify the particles can get onto structures erected by statues, buildings and other people and damage their surfaces. Acid particles damage metal and cause accelerated deterioration of paint and stone. It also lowers the surface of buildings and other structures, such as monuments.
The consequences of this loss can be costly:
- Damaged content that needs to be repaired or replaced.
- Increased maintenance costs and
- Details are lost in stone and metal statues, monuments and tombs.
Indirect Effects of Acid Rain
It’s all about air quality. While acid rain cannot directly harm a person, the sulfur dioxide it produces can cause health problems. Sulfur dioxide particles in the air, in particular, can cause chronic lung problems such as asthma and bronchitis.
In addition, nitrogen oxides, which are responsible for acid rain, promote the formation of ozone in the soil. While ground-level ozone blocks ultraviolet radiation, ground-level ozone causes serious lung problems, such as chronic pneumonia and emphysema.
When acid rain falls at higher elevations, the acid rain creates a dense acid mist that hangs down, impairing vision and irritating the eyes and nose. The acid mist also affects trees and shrubs, turning their leaves brown and dry.
In addition to the effects of acid rain on air quality, acid rain also has a major impact on the balance of the environment. Acid rain falling directly on trees and shrubs can damage them. Acid rain runoff deposits minerals such as aluminum in the soil, which lowers the pH and makes the soil acidic. Acidic soils are harmful to crops and spoilage.
As acid drainage flows into lakes, rivers and oceans, it disrupts the balance of these aquatic ecosystems and causes damage or even death to aquatic organisms. Imbalances in aquatic ecosystems have a negative effect on fisheries.